Seasonality and the Ethanol Blending Margin
In yesterday’s post the relationship between gasoline and ethanol prices was explored. The importance of tracking the difference between gasoline and ethanol prices (Figure 1) as an indicator of the “blending margin” between gasoline and ethanol was highlighted. A positive difference means market blending economics favor ethanol and vice versa. In recent years it has been profitable to blend ethanol in gasoline about 70% of the time (without consideration of the blenders’ tax credit).

It is important to realize that the difference between ethanol and gasoline prices is only an indicator of the true blending margin for ethanol because margins are influenced by more than this difference. Transportation fuel is a blend of ethanol and a number of petroleum products, such as methane, butane, and naptha. The blend of these petroleum products changes seasonally (due to weather and changes in emissions requirements) and as the relative prices of the various products change. Much like the price of feed ingredients in a least-cost feed ration, changes in the price of these other petroleum products can influence the value of ethanol in the blend.
Figure 2 presents some evidence about the seasonal dynamics of the relative value of ethanol in gasoline blends. The data are the same as in Figure 1 but the data are plotted by calendar week in the year rather than as a continuous series. A clear seasonal tendency is present, with ethanol prices low relative to gasoline in the spring and summer and higher in the fall and winter. This tendency is even more obvious in Figure 3, which averages the difference across years for the same week. The bottom line is that ethanol is $0.40 to $0.50 more valuable in gasoline blends in the fall and winter.

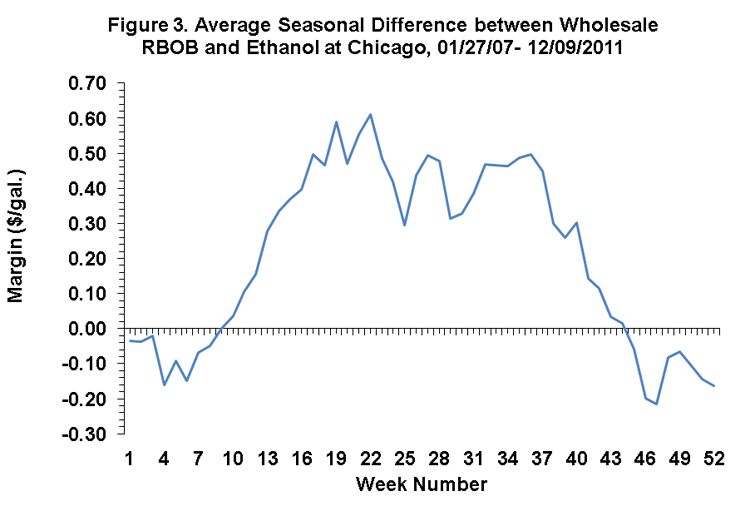
What is the source of this higher seasonal value? It is likely related to the vapor pressure properties of ethanol. Winter gasoline blends throughout much of the U.S. require higher vapor pressure in order to assure engines start in cold conditions and ethanol has a relatively high vapor pressure. However, this is probably not the entire story, as gasoline blending is an amazingly complicated process. Further digging is required.
Disclaimer: We request all readers, electronic media and others follow our citation guidelines when re-posting articles from farmdoc daily. Guidelines are available here. The farmdoc daily website falls under University of Illinois copyright and intellectual property rights. For a detailed statement, please see the University of Illinois Copyright Information and Policies here.







