When It Rains, It Pours: Extreme Precipitation & Nutrient Loss, Part 3
Scientists recently confirmed last year’s lived experience: 2023 was the hottest year on record, and by a lot. The European climate monitor announced that global average temperature last year was 2.66 degrees Fahrenheit warmer as measured against global average temperatures for the pre-industrial base period of 1850-1900 (Copernicus, January 9, 2024; Zhong and Collins, January 9, 2024; Poynting and Rivault, January 9, 2024). In general, warmer air holds more moisture and tends to produce larger precipitation events, or extreme rains; as the planet continues to warm, the more common these events will become (see e.g., NOAA, NCEI, November 18, 2020). Among the many impacts on farming from extreme precipitation are those on nutrient losses from soils and fields. Previously in this series, we covered the basics of nutrient loss and precipitation (farmdoc daily, December 7, 2023) and the impact of extreme precipitation on phosphorus runoff (farmdoc daily, January 4, 2023). Today’s article focuses on the role of precipitation in driving losses of nitrogen.
Background
This discussion focuses on nitrogen lost from the soils of farm fields. As one researcher has written, the “nitrogen cycle is both fascinating and frustrating in its complexity” (Myrold, 2021). What follows is a summary of the scientific understanding about the nitrogen cycle and losses; it is not intended to be a comprehensive or complete scientific explanation of the complex biochemical processes at work. It is, at best, a crash course on the complex topic intended as background, built on the basics drawn from an incredible wealth of research (see also, farmdoc daily, March 10, 2016). The goal is to help support an understanding of the challenging issue that is nutrient losses from farming.
Nitrogen is crucial for life, growth, and development and lack of it is one of the most important limiting factors on plant growth and productivity. Researchers have found that nitrogen cycles through nine forms—no other essential nutrient has as many—and that farming can lose nitrogen in multiple of those forms, much of it in a short time frame (i.e., less than one year). Most of the nitrogen in soil is in the organic form and not available to plants, becoming available to plants relatively slowly. Plant roots take up mostly inorganic nitrate and ammonium forms, both of which exist in the soil naturally and are also added by fertilizer inputs. Humans have drastically increased the total amount of reactive nitrogen that cycles through its various forms and moves through the natural environment since discovering the process for synthesizing ammonia, known as Haber-Bosch, in 1909. Researchers have estimated, however, that a substantial portion of this nitrogen is not consumed by plants, animals, or humans. What isn’t consumed leaches into waterways or is released to the atmosphere; the former is a challenge to water quality, the latter adds to the challenge of climate change (Cao, Lu and Yu, 2018; Robertson and Groffman, 2007; Smil, 2002; Russel and Williams, 1977).
Microorganisms are critical players in the nitrogen cycle. They consume biomass and nitrogen. If the biomass provides plentiful nitrogen, the microorganisms release what they don’t need in an inorganic form (mineralization), but if the biomass does not provide sufficient nitrogen the microorganisms must consume nitrogen from the soil (immobilization). Mineralization increases the nitrogen available to plants; immobilization decreases it. Microorganisms can also produce ammonium from inorganic nitrogen, and when they produce more ammonium in the soil than plants need or can consume, then nitrification can take place. This further conversion takes ammonium to nitrite and then to nitrate forms. It is the nitrate form that is especially mobile and susceptible to being leached. In general, ammonium can be held in the soil, but nitrate is easily dissolved in and transported by water, especially when precipitation exceeds evapotranspiration. Another important note about the ammonium and nitrate forms for farming is that researchers have found plants and microorganisms prefer (or prioritize) applied nitrogen in place of naturally occurring nitrogen. Timing is also an issue. Early in the season, crop growth and nitrogen uptake is low but mineralization is high (especially from applied nitrogen), leaving nitrogen at greater risk of being lost due to precipitation before the growing crop can use it. Finally, the nitrate form can also go through denitrification and be lost to the atmosphere. If it is lost as nitrous oxide, it is a concern because that is a greenhouse gas and may also contribute to the destruction of ozone (see e.g., Myrold, 2021; Fernández, Fabrizzi and Naeve, 2017; Robertson et al., 2013; Dessureault-Rompré et al., 2011; Robertson and Vitousek, 2009; Robertson and Groffman, 2007; Smil, 2002; Cassman et al., 2002; Williams, Hutchinson and Fehsenfeld, 1992).
The complex system of artificial (human-made) drainage is also a critical component of nitrogen losses because it quickly moves water from precipitation and snowmelt out of farm fields. Subsurface tile drainage is prominent in the Midwest for farming soils that are naturally poorly drained. As the system quickly moves water out of fields it inevitably carries with it large quantities of nitrates and other nutrients that are transported to lakes, reservoirs, and natural waterways. Nitrogen fertilizers, even in the ammonia form, are subject to rapid nitrification to nitrate and easily transported from soils and fields by the drainage system. The risk of loss is magnified if the fertilizer is applied in the fall after harvest or too early in the spring because the nitrogen is available to washed out by the weather, carried by water from rain and snowmelt. The science on this is clear and has been consistent for many years (see e.g., Gentry et al., 2023; Li et al. 2022; Pittlekow et al., 2017; Christianson and Harmel, 2015; David, Drinkwater, and McIsaac, 2010; Gentry et al., 2009; Kladivko et al., 1991).
Discussion
The massive increase in synthetic nitrogen fertilizer feeds a “nitrogen cascade” through the environment (Robertson and Groffman, 2007). Within this cascade are vast quantities of excess nitrate that is susceptible to leaching through soils during precipitation events and exported from fields by artificial drainage, degrading water quality from local streams to the Mississippi River and the Gulf of Mexico. Recent research on a decade of surface water quality readings in Wisconsin measured ammonia as an indicator of nitrogen loss because direct measurement of nitrogen itself is uncommon. The research found elevated ammonia levels in surface water from larger rain events as compared to a day with less than half an inch of rain, such that: on a day with one half to one inch of rain, ammonia increased by 39%; on a day with one to two inches of rain, it increased by 56%; and on a day with more than two inches of rain, ammonia increased 59% (Skidmore et al., 2023).
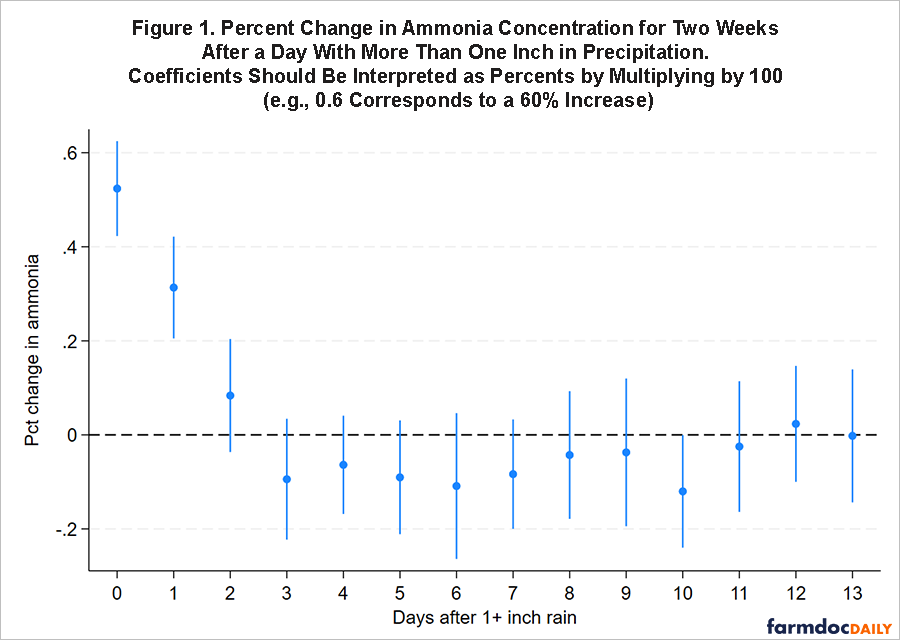
A comparison with phosphorus, discussed previously, may be of value. In general terms, phosphorus attaches to soil particles and moves relatively slowly through ecosystems, usually lost as surface runoff (see e.g., Schneider et al., 2019; farmdoc daily, January 4, 2024). By comparison, nitrogen (especially nitrate) dissolves in water and moves through ecosystems much more rapidly (and usually through drainage). The difference is apparent in water quality readings. By three days after the precipitation event, for example, elevated ammonia levels are no longer evident in water quality readings. Figure 1 illustrates the change in ammonia concentration each day for two weeks after a day with more than one inch of precipitation. Spring and summer rainfall also do not drive higher ammonia levels at the end of the season (October – December). This follows what we expect with a soluble nutrient. It’s extremely responsive, both entering and exiting systems quickly.
The seasonality of nitrogen losses again shows that nitrogen runoff is the worst when precipitation (including snowmelt) occurs on bare ground. Figure 2 illustrates the increase in ammonia in water on days with more than an inch of precipitation by month in which the precipitation takes place. Ammonia spikes are high following rainfall in April and May, which points to the risk of losing recently applied in spring applications before the crop emerges. Ammonia is also very responsive to precipitation in December, when soils are likely frozen (or freezing and thawing). Notably, application on frozen ground or snow-covered ground is restricted in many counties in Wisconsin (e.g., NR 151 Silurian bedrock standard link; Dane County animal waste standard). As discussed in the background, significant losses from November through March are likely due to post-harvest, fall application of nitrogen fertilizer.
Notably for the Corn Belt, only 10% of cropland in Wisconsin is drained by subsurface tile as of 2020 (Wisconsin Discover Farms, 2020). This is significantly less than in other key Midwestern states such as Illinois, Iowa, or Indiana (Wisconsin Discovery Farms, 2017). It also differs in nature, as Wisconsin’s agricultural land includes more hilly terrain than its neighbors to the south (Wisconsin Discovery Farms, 2009). Therefore, these elevated levels of ammonia in surface waters in Wisconsin largely reflect overland runoff rather than from subsurface drainage.
On the upside, the responsive nature of nitrogen also means that efforts to reduce it are more likely to have an impact in the short run. For example, nitrogen levels fall with more EQIP payments (Liu et al., 2022), support and requirements for nutrient management planning (Skidmore et al., 2023), and cover cropping (Hsieh and Gramig, 2023). Better nutrient management practices, such as avoiding fall applications and split applications in the spring, can also have a substantial, positive impact on nitrogen losses (see e.g., Gentry et al., 2023).
Conclusion
Nutrient loss from farm fields presents one of the more perplexing natural resource challenges, and losing nitrogen lays bare and makes clear the complexities of the matter. The critical stage of the nitrogen cycle that makes the nutrient available to plants for growth and production, also makes nitrogen incredibly vulnerable to being leached and lost. Dissolved in water, nitrogen in the nitrate form (and dissolved organic nitrogen) can be carried to waterways and away from plant roots by drainage systems where it degrades water quality and contributes to hypoxic or dead zones. When it rains, it pours; farmers can do nothing about the rain and little to control the nitrogen cycle, but they can manage fertilizer applications to avoid excess nitrogen in the fields at critical times. With climate change and increasing global temperatures, some of the consequences at the local level will be the increasing frequency of extreme precipitation events. Heavy rains on farm fields will magnify the challenges for farmers, crops, and public waters unless farmers adopt practices found to reduce those losses. Public policies can and do support farmers in optimizing nitrogen and land use to meet crop needs and benefit the public, but the scale and scope of the challenge calls for more—more support and more creative, innovative policy options. Recent research reviewed in this series adds valuable knowledge and contributes important perspective.
References
Bateman, E. J., and E. M. Baggs. "Contributions of nitrification and denitrification to N 2 O emissions from soils at different water-filled pore space." Biology and fertility of soils 41 (2005): 379-388. https://link.springer.com/article/10.1007/s00374-005-0858-3.
Cassman, Kenneth G., Achim Dobermann, and Daniel T. Walters. “Agroecosystems, Nitrogen-use Efficiency, and Nitrogen Management.” AMBIO: A Journal of the Human Environment 31, no. 2 (2002): 132-140. https://doi.org/10.1579/0044-7447-31.2.132.
Cao, Peiyu, Chaoqun Lu, and Zhen Yu. "Historical nitrogen fertilizer use in agricultural ecosystems of the contiguous United States during 1850–2015: application rate, timing, and fertilizer types." Earth System Science Data 10, no. 2 (2018): 969-984. https://essd.copernicus.org/articles/10/969/2018/.
Christianson, L. E., and R. D. Harmel. "The MANAGE Drain Load database: Review and compilation of more than fifty years of North American drainage nutrient studies." Agricultural Water Management 159 (2015): 277-289. https://www.sciencedirect.com/science/article/pii/S0378377415300408.
Copernicus Climate Change Service. “Copernicus: 2023 is the hottest year on record, with global temperatures close to 1.5° limit.” January 9, 2024. https://climate.copernicus.eu/copernicus-2023-hottest-year-record.
Coppess, J. "Dead Zones & Drinking Water, Part 2: Why Tile Matters." farmdoc daily (6):47, Department of Agricultural and Consumer Economics, University of Illinois at Urbana-Champaign, March 10, 2016.
David, Mark B., Laurie E. Drinkwater, and Gregory F. McIsaac. "Sources of nitrate yields in the Mississippi River Basin." Journal of environmental quality 39, no. 5 (2010): 1657-1667. https://doi.org/10.2134/jeq2010.0115.
David, Mark B., Lowell E. Gentry, David A. Kovacic, and Karen M. Smith. Nitrogen balance in and export from an agricultural watershed. Vol. 26, no. 4. American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America, 1997. https://doi.org/10.2134/jeq1997.00472425002600040015x.
Dessureault-Rompré, Jacynthe, Bernie J. Zebarth, Alex Georgallas, David L. Burton, and Cynthia A. Grant. "A biophysical water function to predict the response of soil nitrogen mineralization to soil water content." Geoderma 167 (2011): 214-227. https://doi.org/10.1016/j.geoderma.2011.10.005.
Fernández, Fabián G., Karina P. Fabrizzi, and Seth L. Naeve. "Corn and soybean’s season-long in-situ nitrogen mineralization in drained and undrained soils." Nutrient Cycling in Agroecosystems 107 (2017): 33-47. https://link.springer.com/article/10.1007/s10705-016-9810-1.
Gentry, Lowell E., Mark B. David, Frederick E. Below, Todd V. Royer, and Gregory F. McIsaac. "Nitrogen mass balance of a tile‐drained agricultural watershed in east‐central Illinois." Journal of environmental quality 38, no. 5 (2009): 1841-1847. https://doi.org/10.2134/jeq2008.0406.
Hsieh, Hsin-Chieh, and Benjamin M. Gramig. 2023. “Estimating the Impact of Cover Crop Adoption on Ambient Nitrogen Concentration in the Upper Mississippi River Drainage.” Applied Economic Perspectives and Policy 1–18. https://doi.org/10.1002/aepp.13408.
Kladivko, E. J., G. E. Van Scoyoc, E. J. Monke, K. M. Oates, and W. Pask. Pesticide and nutrient movement into subsurface tile drains on a silt loam soil in Indiana. Vol. 20, no. 1. American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America, 1991. https://acsess.onlinelibrary.wiley.com/doi/10.2134/jeq1991.00472425002000010043x.
Li, Ziyi, Kaiyu Guan, Wang Zhou, Bin Peng, Zhenong Jin, Jinyun Tang, Robert F. Grant et al. "Assessing the impacts of pre-growing-season weather conditions on soil nitrogen dynamics and corn productivity in the US Midwest." Field Crops Research 284 (2022): 108563. https://doi.org/10.1016/j.fcr.2022.108563.
Liu, Pengfei, Yu Wang, and Wei Zhang. 2023. “The Influence of the Environmental Quality Incentives Program on Local Water Quality.” American Journal of Agricultural Economics 105(1): 27–51. https://doi.org/10.1111/ajae.12316.
Myrold, David D. “Transformations of Nitrogen.” In Principles and Applications of Soil Microbiology, pp. 385-421. Elsevier, 2021. https://www.sciencedirect.com/book/9780128202029/principles-and-applications-of-soil-microbiology.
National Oceanic and Atmospheric Administration, National Centers for Environmental Information. “A Warming Earth is Also a Wetter Earth.” November 18, 2020. https://www.ncei.noaa.gov/news/warming-earth-also-wetter-earth.
Paul, K. I., P. J. Polglase, A. M. O'connell, J. C. Carlyle, P. J. Smethurst, and P. K. Khanna. "Defining the relation between soil water content and net nitrogen mineralization." European journal of soil science 54, no. 1 (2003): 39-48. https://doi.org/10.1046/j.1365-2389.2003.00502.x.
Pittelkow, Cameron M., Matthew W. Clover, Robert G. Hoeft, Emerson D. Nafziger, Jeffery J. Warren, Lisa C. Gonzini, and Kristin D. Greer. "Tile drainage nitrate losses and corn yield response to fall and spring nitrogen management." Journal of environmental quality 46, no. 5 (2017): 1057-1064. https://doi.org/10.2134/jeq2017.03.0109.
Poynting, Mark and Erwan Rivault. “2023 confirmed as world’s hottest year on record.” BBC.com. January 9, 2024. https://www.bbc.com/news/science-environment-67861954.
Robertson, G. Philip, Tom W. Bruulsema, Ron J. Gehl, David Kanter, Denise L. Mauzerall, C. Alan Rotz, and Candiss O. Williams. "Nitrogen–climate interactions in US agriculture." Biogeochemistry 114 (2013): 41-70. https://link.springer.com/article/10.1007/s10533-012-9802-4.
Robertson, G. Philip, and Peter M. Vitousek. "Nitrogen in agriculture: balancing the cost of an essential resource." Annual review of environment and resources 34 (2009): 97-125. https://doi.org/10.1146/annurev.environ.032108.105046.
Robertson, G. Philip, and P. M. Groffman. "Nitrogen transformations." In Soil microbiology, ecology and biochemistry, pp. 341-364. Academic Press, 2007.
Russel, Darrell A., and Gerald G. Williams. "History of chemical fertilizer development." Soil Science Society of America Journal 41, no. 2 (1977): 260-265. https://doi.org/10.2136/sssaj1977.03615995004100020020x.
Skidmore, M. and J. Coppess. "When It Rains, It Pours: Extreme Precipitation & Nutrient Loss, Part 2." farmdoc daily (14):3, Department of Agricultural and Consumer Economics, University of Illinois at Urbana-Champaign, January 4, 2024.
Skidmore, M. and J. Coppess. "When It Rains, It Pours: Extreme Precipitation & Nutrient Loss, Part 1." farmdoc daily (13):222, Department of Agricultural and Consumer Economics, University of Illinois at Urbana-Champaign, December 7, 2023.
Skidmore, Marin, Tihitina Andarge, and Jeremy Foltz. 2023. “Effectiveness of Local Regulations on Nonpoint Source Pollution: Evidence from Wisconsin Dairy Farms.” American Journal of Agricultural Economics 105(5): 1333–1364. https://doi.org/10.1111/ajae.12388.
Skidmore, Marin, Tihitina Andarge, and Jeremy Foltz. 2023. “The Impact of Extreme Precipitation on Nutrient Runoff.” Journal of the Agricultural and Applied Economics Association. 1–17. https://doi.org/10.1002/jaa2.90
Smil, Vaclav. “Nitrogen and Food Production: Proteins for Human Diets.” AMBIO: A Journal of the Human Environment 31, no. 2 (2002): 126-131. https://doi.org/10.1579/0044-7447-31.2.126.
Williams, E. J., G. L. Hutchinson, and F. C. Fehsenfeld. “NOx and N2O Emissions from Soil.” Global Biogeochemical Cycles 6, no. 4 (1992): 351-388.
Zhong, Raymond and Keith Collins. “See How 2023 Shattered Records to Become the Hottest Year.” The New York Times. January 9, 2024. https://www.nytimes.com/2024/01/09/climate/2023-warmest-year-record.html.
Disclaimer: We request all readers, electronic media and others follow our citation guidelines when re-posting articles from farmdoc daily. Guidelines are available here. The farmdoc daily website falls under University of Illinois copyright and intellectual property rights. For a detailed statement, please see the University of Illinois Copyright Information and Policies here.